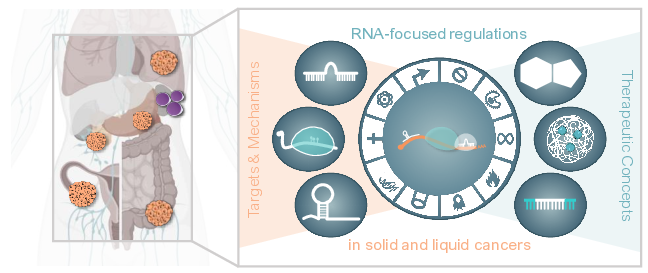
The development of novel therapeutics is a tedious endeavor that requires interdisciplinary research approaches and industrial partners in the long-run. However, academic research on the pathophysiological and molecular mechanisms underlying ncRNA and RBP function(s) in cancer is essential to initiate the pre-clinical development of R3oGE-directed treatment concepts. This research will reveal candidate targets, sets the stage for their pre-clinical evaluation, andprovides the rationale for exploring novel therapeutic strategies, which in most cases originate in academic research. Scientists participating in the here proposed research consortium have contributed to these research foci of the RU5433.
Hallmark studies by the Heckl/Klusmann labs identified diseases modulating ncRNAs in leukemia – including microRNAs (miRNAs; miR) as well as lncRNAs (long non-coding RNAs). These in part ERC-funded (Klusmann) studies provide a comprehensive view on the ncRNA landscape of hematopoiesis as well as leukemia. This was instrumental to unravel the role, underlying pathophysiological mechanisms and therapeutic potential of miRNAs and lncRNAs in leukemia. Complementary research by the Aigner, Hämmerle and Hüttelmaier labs provided essential insights on the role and target potential of ncRNAs in solid cancers. The Hüttelmaier group developed techniques and analysis pipelines to reveal conserved functions andregulatory miRNA-mRNA networks in neuroblastoma and thyroid carcinomas. Profiting from ample expertise of the Aigner lab on nanoparticle formulations for the delivery of nucleic acid therapeutics in cancer models, joined studies with the Hüttelmaier labs recently reported the first in vivo feasibility study demonstrating anti-tumor activity of circular miR RNA decoysdelivered via polymeric nanoparticles. The Hämmerle lab contributed to the characterization of lncRNAs in cancer, specifically lncRNAs serving roles in pancreatic cancer, was involved in exploiting the therapeutic value of ncRNAs in tumor therapy, and deciphered the interplay of lncRNAs and pro-oncogenic RBPs in solid cancer.
The role of RBPs in leukemia and solid cancers is the main research interest of the Bley, Höll and Hüttelmaier labs. The Höll group reported mechanisms underlying the role of RBPs in leukemia, and investigated RBP-RNA interaction networks in cancer models. Conserved pathophysiological mechanisms and therapeutic potential of oncofetal RBPs in solid cancers is the core research topic of the closely interacting Bley (née Stöhr) and Hüttelmaier groups. Recently, these work groups teamed up with the Sinz lab providing essential mass spectrometry expertise to decipher RBP-protein interactions networks as well as studying post-translational protein modifications. One major contribution of these joined efforts is the characterization of conserved N6-methyladenosine (m6A) and largely miRNA-dependent mechanisms underlying the RBP-directed stimulation of oncogene expression, cancer cell migration, invasion, and metastasis. Next to a striking potential of oncofetal RBPs in cancer diagnosis, the Hüttelmaier and Bley groups also begun to explore the therapeutic prospects of inhibiting RBPs (RBPi). These studies demonstrated anti-tumor activity of RBPi by small molecules, and synergies of RBPi with kinase inhibition. The development of novel and improvement of prior reported RBP inhibitorsessentially relieson interdisciplinary structure-activity-relation (SAR) guided approaches. To this end, the Hüttelmaier lab teamed up with theBalbach and Sippl labs. These provide complementary expertise in the biophysical characterization of protein folding and protein-ligand interaction by NMR (Balbach) as well as the design and synthesis of PROTACs (proteolysis targeting chimera) as well as small molecule inhibitors (Sippl). These collaborative studies already led to RBPi compounds with superior anti-tumor activity (pending patents: EP20211807.1; EP20159945.3).